The Ministry of Health Malaysia and the National Pharmaceutical Regulatory Agency (NPRA) has warned the public to refrain from purchasing and using the following cosmetic products as they have been detected to contain toxins.
The products include:
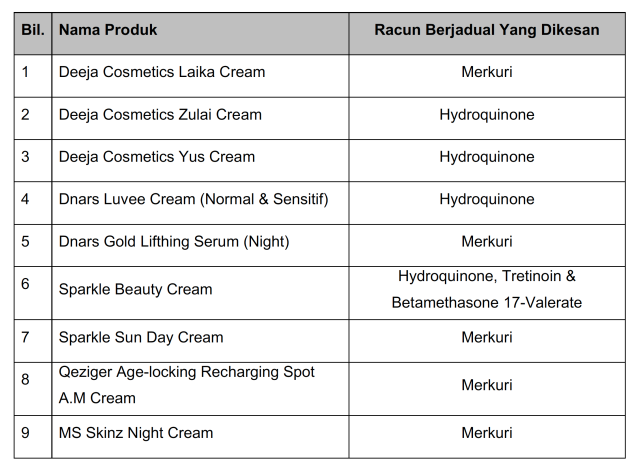
- Deeja Cosmetics (Laika Cream, Zulai Cream, Yus Cream)
- Dnars Luvee Cream (Normal & Sensitive) & Dnars Gold Lifting Serum (Night)
- Sparkle Beauty Cream and Sun Day Cream
- Qeziger Age-locking Recharging Spot A.M Cream
- MS Skinz Night Cream


The senior director of the Ministry of Health has notified to revoke the products that are involved, which means that they are no longer allowed for sale in Malaysia.
What Are Hydroquinone, Tretinoin, Betamethasone 17-valerate And Mercury?
Why are these four substances labelled as toxins in the cosmetics products and what exactly are they?
According to Health Director-general Datuk Dr Noor Hisham Abdullah, the products that contain hydroquinone, tretinoin and betamethasone 17-valerate are drugs that need to be registered with the Drug Control Authority (PBKD) and are only allowed to use under the advice of health professionals.
Without the supervision of health professionals, the usage of products containing these substances will cause unwanted consequences such as red rashes on the skin, change in the colour of skin tone, skin irritation, pain and acne problem.
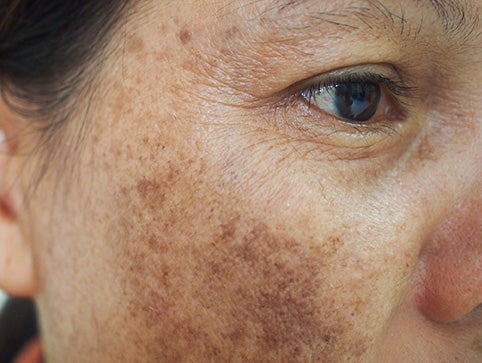
On the other hand, Mercury is banned for use in cosmetics products as it will jeopardise the user’s health. This is because after it is absorbed into the body, it will damage the nervous system as well as the kidney. Rashes and skin irritation can also happen if you use cosmetics products that consist of mercury. Additionally, the mercury will disrupt the brain development of young children and even unborn babies.
Reminder for the product seller, product distributor and the public from MOH
Both the seller and distributor of the products involved are considered to be violating Malaysia’s Control of Drugs and Cosmetics Regulations 1984 and need to discontinue the sale of the products immediately.
Individuals who fail to obey the law will be charged a fine not exceeding RM25,000 or imprisonment not more than three years or both for the first offence and a fine not exceeding RM50,000 or imprisonment not more than five years or both for subsequent offences.
Companies who violate this the law, on the other hand, can be fined up to RM50,000 for the first offence and fined up to RM100,000 for subsequent offences.

For the current product users, MOH urges to stop using them and to seek advice from health professionals if they encounter any discomfort or side effects.
Please ensure to check the list of hazardous products on the NPRA website to ensure that your purchase items that are safe to be used or download the “NPRA Product Status” application from the Google Play Store.
Take care of your skin and your health!
Also Read: “Sausage Is Not Real Meat”, MOH Urges Public To Reduce Sausage Intake